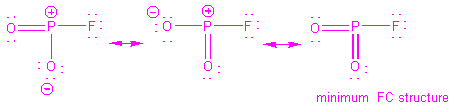
Procedure for applying VSEPR to determine molecular shape:

| # Electron Groups | Group Distribution | Stereochemistry | Visual Description | Examples |
|---|---|---|---|---|
| 2 | linear | linear | BeH2 (gas) | |
| 3 | trigonal planar | 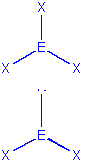 |
trigonal planar
bent |
BCl3, COCl2
SnCl2, SO2, NOCl |
| 4 | tetrahedral | 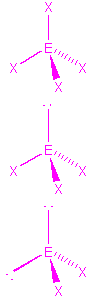 |
tetrahedral
trigonal pyramidal
bent |
CH4, POCl3, SO2Cl2
NH3, SO32-, SOCl2
H2O, ClO2- |
| 5 | trigonal bipyramidal
Note: LP go in
|
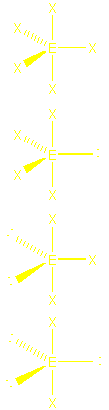 |
trigonal bipyramidal
120o, 90o see-saw
T
linear 180o |
PF5
SF4, IO2F2-
BrF3
XeF2 |
| 6 | octahedral | 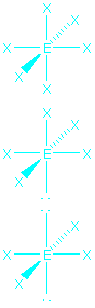 |
octahedral, 90o
square pyramid, 90o
square planar, 90o |
SF6
BrF5
IF4- |