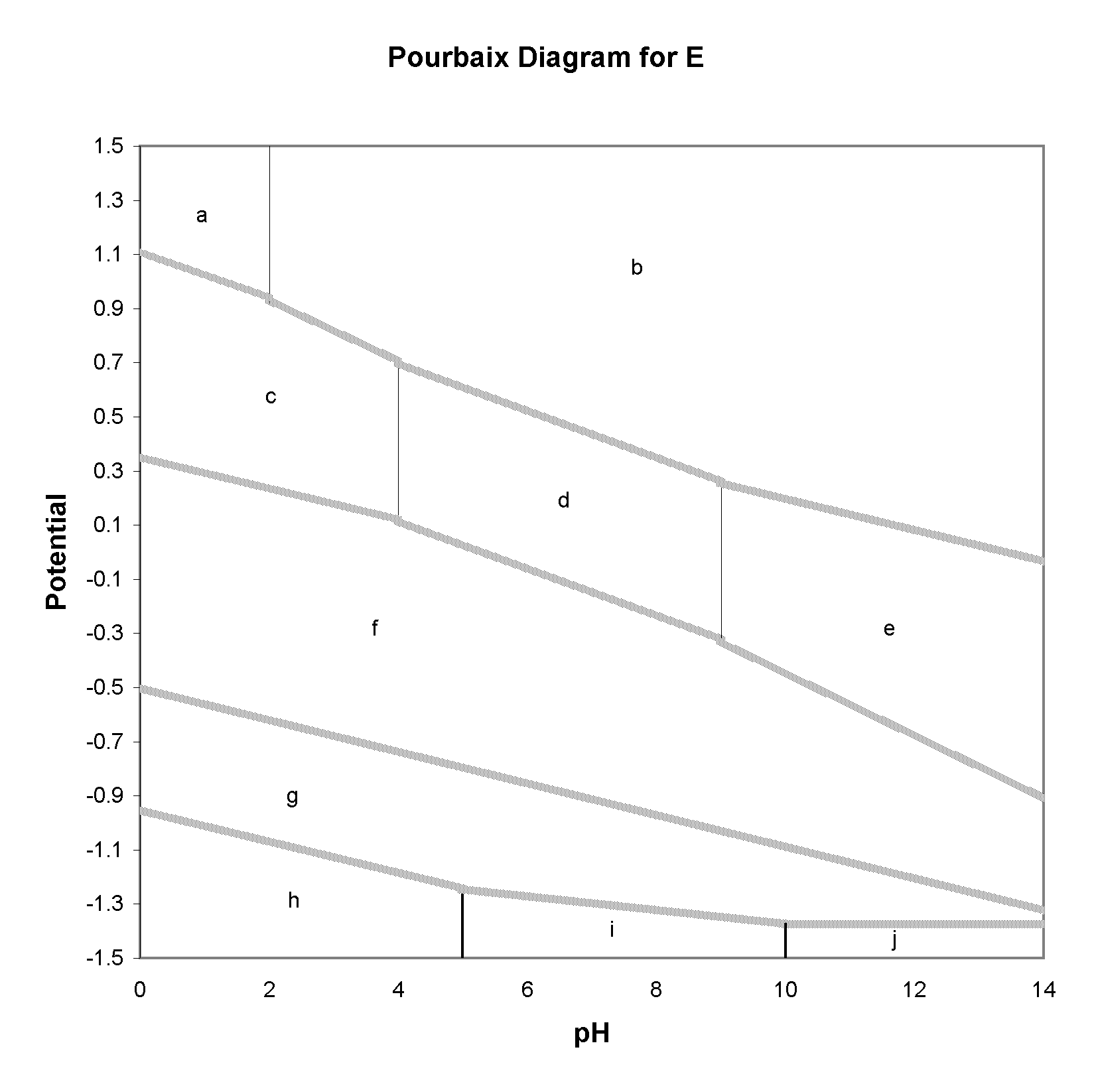
1. (10 points) The Pourbaix Diagram for a hypothetical element, E, is shown below. Various regions are labelled with letters. Below the diagram are listed species formed by E under various conditions of pH and potential in aqueous solut ion. Below the formula for each species, write the letter of the region in which it should occur in the diagram.

| E | H2E | HEO4- | H2EO3 | EO42- | HE- | EO | EO32- | HEO3- | E2- |
| g | h | a | c | b | i | f | e | d | j |
2. (5 points) Write a balanced half reaction for reduction of H2EO3 at pH 2, and estimate the standard reduction potential.
H2EO3 + 2H+ + 2e ---> EO + 2H2O
e o = 0.35 V (value of potential at pH = 0).
3. (5 points) Develop Lewis structures for EO32- and EO42-, and determine stereochemistry at E.
Structures:
![]()
In EO32-, stereochemistry at E is trigonal pyramidal (4 electron groups, one lone pair and three attaching atoms). In EO42-, it is tetrahedral (4 electron groups, all attaching atoms)