1. (5 points) Complete the s and p blocks of the attached blank periodic table.
2. (10 points) Using the first row
as a model, complete the following table:
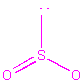
| Formula | Lewis Structure | Total Number of resonance forms | Number of electron groups around central atom | electron group distribution | molecular shape | stereochemical drawing |
| SO2 | 3 | 3 | trigonal planar | bent |  |
|
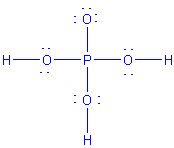
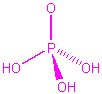
| H3PO4 |  |
2 | 4 | tetrahedral | tetrahedral |  |
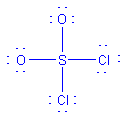
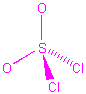
| SO2Cl2 |  |
4 | 4 | tetrahedral | tetrahedral |  |
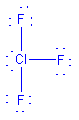
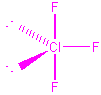
| ClF3 |  |
1 | 5 | trigonal bipyramid | T-shaped |  |
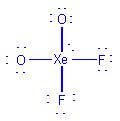
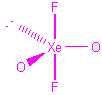
| XeO2F2 |  |
4 | 5 | trigonal bipyramid | see-saw |  |
3. (5 points) Predict the form in which each cation will exist in aqueous solution of pH about 7.
Fe2+: Z2/r = 4/92 = 0.043. The cation is moderately acidic. It will exist as the neutral hydroxide, Fe(OH)2(H2O)4, which will precipitate as a solid.
As5+: Z2/r = 25/60 = 0.42. The cation is very strongly acidic. It will exist as the oxoacid (H3AsO4) or hydroxoanions (H2AsO4-, etc.).
S4+: Z2/r = = 16/51 = 0.31. The cation is very strongly acidic. It will exist as the oxoacid (H2SO3) or hydroxoanion (HSO3-).