Oxide Structures
Radius Ratio Approach
-
For the cation of interest, determine the formula of the
oxide.
-
Calculate rcat/ran
-
Predict the total coordination number (TCN) of the least
abundant atom (usually the cation). This can be done using the following
guidelines:
| radius ratio range |
CN of cation |
| 0.155-0.224 |
3 |
| 0.224-0.414 |
4 |
| 0.414-0.732 |
6 |
| > 0.732 |
8 |
-
If the cation has lone pairs, subtract the number of lone
pairs from the TCN. This gives the number of oxygen atoms that will fit
in the coordination sphere of the cation. We will call this the CN of the
cation. (The number of lone pairs must be subtracted from the TCN because
each lone pair takes up one position in the coordination sphere.)
CN = TCN - Number of cation lone pairs
-
Calculate the CN of the most abundant (terminal) atom (oxide
anion for our present purposes), basing the calculation on the CN (not
the TCN) of the least abundant atom. This atom has lower CN, and will do
the linking.
CN(cation) x Number of cations = CN(oxide anion) x Number
of oxide anions If CN of terminal atom
= 1, monomeric formula units
1-2, oligomeric or 1-D polymer
> 2, polymer or ionic.
NOTE: For a covalent compound, the CN of oxygen should
be at least 1 and at most equal to the normal number of covalent bonds
formed by oxygen, which is 2. It is VERY UNLIKELY that the CN of oxygen
would exceed 2 in a covalent cpd. The reasons for these limits are as follows:
An average CN less than 1 implies that at least one of
the oxygens is not bonded (i.e., has CN of zero)! This is clearly ridiculous.
If the assumed CN of the cation produces an oxygen CN less than 1, increase
the CN of the cation to the next higher reasonable value (e.g., if the
original CN is 3, move up to 4). Then recalculate the CN of oxygen. It
should now be equal to or greater than 1.
An average CN greater than the normal number of covalent
bonds for oxygen (2) is unlikely because it would cause some of the oxygens
to have positive formal charges. Positive formal charges are unfavorable
for oxygen.
If CN of oxygen exceeds its normal number of covalent
bonds (2) in a compound that must be covalent (the cation and oxygen are
close in the PT), decrease the coordination number of the least abundant
atom (cation) to the next reasonable value (e.g., if original CN is 6,
drop down to 4 ). Then recalculate the CN of oxygen. It should now be less
than or equal to 2.
-
Predict a structure that is consistent with:
-
The stoichiometry of the element oxide
-
The coordination numbers determined above
-
The usual rules governing Lewis structures.
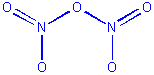
Example: N2O5
We recognize that this should be a covalent compound.
r(N5+) = 27 pm; r(O2-) = 126 pm,
therefore, r+/r- = 0.214
From the value of r+/r-, we conclude that the TCN of the
least abundant atom, N, is 3:
Because N is in its highest oxidation state, there are
no lone pairs. Therefore
CN(N) = 3
This enables us to calculate the CN of the oxide ion:
Thus CN(O) = 2 x 3/5 = 6/5 It is best to leave this
number in fractional form, rather than express it in decimal form.
We interpret the fraction 6/5 to mean that 6 links
(numerator) are made by 5 atoms (denominator). In general, for CN(O) =
x/y, we conclude that x links are made by y atoms.
A link is defined as an attachment between 2 atoms. It
is NOT the same thing as a bond. We will consider a double bond or a triple
bond between two atoms to be one link between them.
Further, we note that the difference between the number
of links (numerator) and the number of atoms (denominator) is the number
of atoms making 2 links:
# links - # atoms = # atoms making 2 links!
Thus in N2O5, 5 oxygen atoms make
6 links; and one oxygen atom (6 - 5) makes 2 links. The other 4 make one
link each.
Structure:
1 O atom has 2 links, 4 have 1 link
4 terminal and 1 bridging O
We begin by placing the 2-link oxygen in the center, and
attaching it to two nitrogens. We then put two 1-link oxygens on each nitrogen:
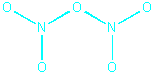
This structure satisfies the stoichiometry (3 Os per
2 Ns) and the coordination number requirements, but is not an acceptable
Lewis structure because although it contains the correct number of electrons,
the Ns only have 6 electrons each. We can fix this by using lone pairs
on two terminal oxgyens to form double bonds to N: