CHEM 281, Chapter 7. HW 7
1. Define the following terms: (a) protonic bridge; (b) hydridic bridge.
2. Define the following terms: (a) borane; (b) Three-center-two electron
bond.
3. Which of the following isotopes can be studied by nuclear magnetic resonance:
carbon-12, oxygen-16, oxygen-17? Explain your answer!
4. Assign the peaks in the 1H NMR of the following compound. Explain the splitting of peaks in the spectrum.
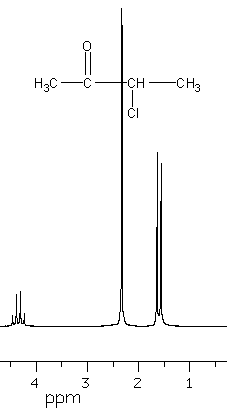
5. Explain why hydrogen is not placed with the alkali metals or
halogens in the periodic table.
6. Is the reaction of dihydrogen with dinitrogen to produce ammonia
entropy or enthalpy driven? Do not consult data tables. Explain your reasoning.
7. Write chemical equations for the reaction of
(a) potassium hydrogen carbonate on heating
(b) ethyne, HCº CH, with dihydrogen
(c) lead(IV) oxide with hydrogen gas on heating
(d) calcium hydride and water
8. Construct a theoretical enthalpy cycle (similar to Figure 7.3) for
the formation of ammonia from its elements. Obtain bond energy information and
the standard enthalpy of formation of ammonia from the data tables in the
Appendices. Compare your diagram with that in Figure 7.3 and comment on the
differences.
9. Which of the following elements is likely ionic, metallic, or
covalent hydride, or non stable hydride:
(a) chromium;
(b) silver;
(c) phosphorus;
(d) potassium.
10. Construct a diagram similar to that of Figure 7.16 to explain why
the hydroxide ion has a very high electrical conductivity.
11. Calculate an approximate value for the bond energy for the
bridging B-H bond in diborane using the data tables in the Appendices. In
comparison with the normal B-H bond energy, what does this suggest about the
bond order? Is this result compatible with the bond order (per bond) deduced
from the molecular orbital diagram (Figure 7.10)?
12. The hydride ion is sometimes considered as similar to a halide
ion; for example, the lattice energies of sodium hydride and sodium chloride
are -808 kJ-mol-1 and -788 kJ-mol-1 respectively,However, the enthalpy of
formation of sodium hydride is much less than that of a sodium halide, such as
sodium chloride. Use the data tables in the Appendices to calculate enthalpy of
formation values for the two compounds and identify the factor(s)that cause the
values to be so different.
13. Instead of the commonly used reaction of hydrocarbons with air as a
high performance aircraft fuel, it was once proposed to use diborane with air.
If
DHcombustion(B2H6(g)) = -2165 kj-mol-1
DHcombustion(C2H6(g)) = - 1560 kj-mol-1
the comparative energy per gram of diborane compared with per gram of ethane. Use the data in the Appendices to calculate the entropy change in each reaction. Suggest why the entropy of combustion values are so different. What would be some practical disadvantage of using diborane?
14.Hydrogen gas has been proposed as the the best fuel for the 21st
century. However, a company in Florida has developed AquaFuel" as an
alternative. This gas mixture is formed by passing a high current through water
using carbon electrodes.
What are the likely gaseous products of electrolysis? Write a balanced
molecular equation for the process. Write a balanced molecular equation for the
combustion of mixture. Calculate the energy released per mole of gas mixture
and compare it with the energy release per mole of hydrogen gas combustion.