Ethers have two alkyl or aryl groups attached to an oxygen atom. The general structure of an ether is R-O-R', where R' again is any alkyl group. R' may or may not be identical to R. Ether = R-O-R
Thiols are a family of compounds that contain the sulfhydryl group (-SH). Thiol = R-SH
An alcohol molecule can be compared to a water molecule. In an alcohol molecule, the hydroxyl oxygen and the two atoms bonded to it are all in the same plane and have a bond angle of approximately 104o. This is very similar to a water molecule.
Water = H-O-H Alcohol = R-O-H
The hydroxyl group is polar.
There is a partial negative charge (d-) on the
oxygen atom and a partial positive charge (d+)
on the hydrogen of the hydroxyl group.
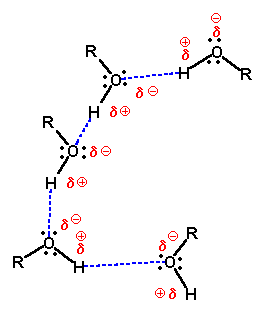
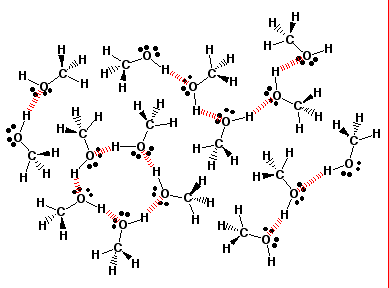
Alcohols boil at higher temperature than alkanes of similar MW. The higher boiling point is due to hydrogen bonding.
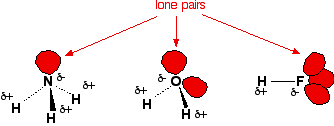
If hydrogen is joined to any of the three most electronegative elements, fluorine, oxygen or nitrogen by a covalent bond, the bond is polar. The bonding electrons are shared so unevenly that the exposed proton at the hydrogen end is strongly attracted to non-bonding pairs of electrons on other molecules. This is called hydrogen bonding. It is the strongest of the intermolecular forces.
A hydrogen bond is an intermolecular attraction in which
a hydrogen atom that is bonded to an electronegative atom, and therefore
has a partial positive charge, is attracted to an unshared electron pair
on another small electronegative atom.
 |
 |
|
 |
 |
|
A general rule of solubility is "Like molecules dissolve like molecules". Polar substances are soluble in polar solvents. Nonpolar substances are soluble in nonpolar solvents.
Alcohols are miscible in water the following "Rule of Thumb" is followed. An organic compound is soluble in water if the Carbon to Oxygen ratio in the compound does not exceed 4:1.
| Compound | Formula | Solubility: g/100 g H2O |
| Methanol | CH3OH | Infinitely soluble |
| Ethanol | C2H5OH | Infinitely soluble |
| Propanol | C3H7OH | Infinitely soluble |
| Butanol | C4H9OH | 8.88 |
| Pentanol | C5H11OH | 2.73 |
| Hexanol | C6H13OH | 0.602 |
| Heptanol | C7H15OH | 0.174 |
The hydroxyl group area of the alcohol is polar and therefore hydrophilic while the carbon chain portion is nonpolar and hydrophobic. As the carbon chain becomes longer, the molecule increasingly becomes overall more nonpolar and therefore less soluble in the polar water.
Methanol CH3-OH
Ethanol CH3-CH2-OH
1-Propanol CH3-CH2-CH2-OH
1-Butanol CH3-CH2-CH2-CH2-OH
1-Pentanol CH3-CH2-CH2-CH2-CH2-OH
1-Hexanol CH3-CH2-CH2-CH2-CH2-CH2-OH
1-Heptanol CH3-CH2-CH2-CH2-CH2-CH2-CH2-OH
Alcohol Nomenclature
1. Determine the name of the parent compound, the longest
continuous carbon chain containing the -OH group.
2. When the -OH group is present in a hydrocarbon, the
-e ending of the alkane, alkene and alkyne is replace by -ol.
3. The numbering is assigned such that the -OH group
gets the lowest possible number.
4. Alcohols containing two hydroxyl groups are named
-diols. Those bearing three are called -triols. A number giving the position
of each of the hydroxyl groups is needed in theses cases.
5. If an alkene and an alkyne also contain the -OH group,
the name is expressed as:
if an alkene: ___-en___ol [first blank
gives position of double bond, second blank the position of -OH group]
if an alkene: ___-yn___ol [first blank gives position
of triple bond, second blank the position of -OH group]
| methanol CH3-OH | 2-propanol |
| 2-methyl-1-butanol |
2-methyl-2-butanol |
| 2-phenylethanol |
cyclohexanol |
| 2-propen-1-ol |
3-butyn-2-ol |
The common names for alcohols are derived from the alkyl group corresponding to the parent compound.
|
|
CH3CH2OH |
|
Common Alcohols (The following information
was obtained at, http://www.bartleby.com.)
Methanol is a colorless, toxic, flammable liquid, CH3OH,
used as an antifreeze, a general solvent, a fuel, and a denaturant for
ethyl alcohol. Also called carbinol, methyl alcohol, wood alcohol, wood
spirits.
Ethanol is a colorless volatile flammable liquid, C2H5OH,
synthesized or obtained by fermentation of sugars and starches and widely
used, either pure or denatured, as a solvent and in drugs, cleaning solutions,
explosives, and intoxicating
beverages. Also called ethanol, ethyl alcohol, grain
alcohol.
2-Propanol (isopropyl alcohol) is a clear, colorless, flammable, mobile liquid, (CH3)2CHOH, used in antifreeze compounds, in lotions and cosmetics, and as a solvent for gums, shellac, and essential oils. Rubbing alcohols is a mixture usually consisting of 70 percent isopropyl or absolute alcohol, applied externally to relieve muscle and joint pain.
1,2-Ethanediol (ethylene glycol) is a colorless syrupy alcohol, HOCH2CH2OH, used as an antifreeze in cooling and heating systems.
1,2,3-Propanetriol (glycerol) is a syrupy, sweet, colorless or yellowish liquid, C3H8O3, obtained from fats and oils as a byproduct of saponification and used as a solvent, an antifreeze, a plasticizer, and a sweetener and in the manufacture of dynamite, cosmetics, liquid soaps, inks, and lubricants. cosmetics, pharmaceuticals, and lubricants. Glycerol is a by-product of the hydrolysis of fats.
Glycerol
Classification of Alcohols
Alcohols are classified depending on the number of alkyl
groups attached to the carbinol carbon.
1o alcohol: -OH bonded to a carbon which is
bonded to one other carbon atom.
2o alcohol: -OH bonded to a carbon which is
bonded to two other carbon atoms.
3o alcohol: -OH bonded to a carbon which is
bonded to three other carbon atoms.
|
|
|
|
|
|
|
|
Alcohol Reactions
1. Preparation
a. Hydration of alkenes
In the presence of acid, water adds to an alkene double bond.


b. Hydrogenation (reduction)
of aldehydes and ketones
2. Dehydration: An Elimination Reaction
Dehydration of an alcohol is
the opposite of the hydration of an alkene.
R-CH2-CH2-OH + H2SO4 ---> R-CH=CH2
3. Oxidation Reactions
In organic terminology:
Oxidation is
1. The addition of oxygen to a molecule.
2. The removal of hydrogen from a
molecule.
Reduction is
1. The removal of oxygen from a molecule.
2. The addition of hydrogen to a molecule.
| Oxidation of a primary alcohol yields an aldehyde.
1o alcohol ------> Aldehyde |
| Oxidation of a secondary alcohol yields an ketone.
2o alcohol ------> Ketone |
| Tertiary alcohols do not undergo oxidation.
3o alcohol ------> NR |
|
Methanol -----> Methanal |
|
2-propanol ----> Acetone |
Note:
1. Every time an oxidation is carried out, there must
also be a reduction.
2. In the complete sense, an oxidation is a loss of electrons
and a reduction is a gain of electrons.
3. Reduction is the addition of hydrogen and or the loss
of oxygen.
Oxidation of an alkene by reaction with dilute aqueous
solution of KMnO4 produces a dihydroxy alcohol.
Ethylene + KMnO4 --> ethylene glycol (used
in antifreeze)
Oxidation and Reduction in Living Systems
Oil Rig
| Oxidation
Involves Loss of electrons |
Reduction
Involves Gain of electrons |
Oxidation: Ago ---> Ag1+ + e1- Reduction: Bro + e1- ---> Br1-
Oxidation
1. The addition of oxygen to a molecule.
2. The removal of hydrogen from a
molecule.
Reduction
1. The removal of oxygen from a molecule.
2. The addition of hydrogen to a molecule.
Phenol
Other common phenols are: Thymol [2-isopropyl-5-methylphenol]
(mint; used in flavoring)
Carvarcol [5-isopropyl-2-methylphenol] (savory)
Butylated hydroxy toluene [2,6-di-tert-butyl-p-cresol](BHT; preservative)
Methyl salicylate
Urushiols are dihydroxy phenols found in poision ivy and poision oak.
Urushiols are oils which are secreted on the surface of the plant's leaves
and are easily transferred to clothing and skin when the leaves are touched.
These are the guilty compounds which cause the rash!
Ethers
R-CH2-R R-O-R
H-O-H
Alkane
Ether Water
Polarity of oxygen gives it a solubility with alcohols
due to hydrogen bonding.
Alkane nature gives them solubility with hydrocarbons.
Ether Nomenclature
Name the two groups that are attached to oxygen, and
follow these names by the word ether.
Symmetrical ethers have both groups identical.
Methyl ether or dimethyl ether = CH3-O-CH3
Ethyl ether or diethyl ether = CH3CH2-O-CH2CH3
Phenyl ether = Ar-O-Ar or
Ph-O-Ph
Unsymmetrical ethers have different alkyl groups.
Ethyl methyl ether = CH3CH2-O-CH3
Phenyl propyl ether = Phr-O-CH(CH3)2
The simplest aryl alkyl ether has the special name of anisole. Ph-O-CH3
Thiols
Compounds which contain the sulfhydryl group (-SH) and
have the general formula of R-SH.
trans-2-butene-1-thiol (North American skunk spray)
Thiols undergo oxidation to yield a disulfide bond (-S-S-).
This type of bonding is one of that helps give proteins their structure.
2 R-SH ---------> R-S-S-R
thiol
disulfide
Thiol Nomenclature
The rules for naming thiols are exactly like those for
alcohols, except that the suffix -ol is replaced by -thiol.